Press Release
Galectin Therapeutics, Inc. Announces Positive Preliminary Results from Phase 1b Clinical Trial of GR-MD-02 and KEYTRUDA® in Advanced Melanoma and Expansion of the Trial
- Combination immunotherapy of GR-MD-02 and KEYTRUDA for all cohorts reported shows an Objective Response Rate of 50% (seven of fourteen patients).
- The published response rate of KEYTRUDA alone is 33% in melanoma
- Providence Portland principal investigator, Dr. Brendan Curti, encouraged by the results and plans to add more patients to the study
- Both Advanced Melanoma and Head and Neck Cancer being studied
NORCROSS, Ga.,
|
|||||
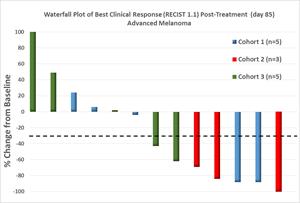
When aggregated with the cohorts previously reported, the data shows a 50% objective response rate in advanced melanoma with GR-MD-02 in combination with KEYTRUDA, and a significant decrease in the frequency of suppressive myeloid-derived suppressor cells (MDSC) following treatment in the responding patients (on day 85 post-treatment) was observed. The published data on KEYTRUDA alone have shown an objective response rate of 33% in this patient population.
Fourteen advanced melanoma patients across three dose cohorts now have Objective Response Rate (ORR) and Disease Control Rate (DCR) data. Six patients in cohort 3 (8 mg/kg GR-MD-02) have now been added to the three patients in cohort 2 (4 mg/kg GR-MD-02) and the five patients in cohort 1 (2 mg/kg GR-MD-02). Cohorts 1 and 3 each had two patients with an objective response. All three patients in cohort 2 had an objective response.
Generally, the
In addition to the fourteen advanced melanoma patients, six patients with head and neck cancer were enrolled in this phase 1b trial with a 33% objective response rate and a 67% disease control rate. Dr. Curti states “the response rates observed overall in advanced melanoma and head and neck cancer patients were better than expected with KEYTRUDA alone and are the basis for moving forward with both tumor types, particularly given the low response rates of anti-PD-1 monotherapy in head and neck cancer. There is a significant clinical need for better options for these patients and our initial objective response rates were encouraging enough to warrant inclusion of additional patients to help determine whether we should also pursue these challenging patient populations in a phase 2 trial. Taken together with the observed favorable safety and tolerability of the combination, these results provide a compelling rationale to move forward with this approach.” Given that all three melanoma patients (100%) were responders at 4 mg/kg dose, the investigators plan to continue the trial with expansion of the 4 mg/kg GR-MD-02 and KEYTRUDA cohort to include additional advanced melanoma patients and additional head and neck cancer patients.
“In addition to the encouraging clinical responses seen thus far, we continue to make progress on identifying immunological biomarkers that correlate with favorable responses,” said William L. Redmond, Ph.D., Associate Member,
“Galectin Therapeutics is very pleased with our continuing collaboration with
Additional information about this clinical trial may be found at: www.clinicaltrials.gov/ct2/show/NCT02575404
About GR-MD-02
GR-MD-02 is a complex carbohydrate drug that targets galectin-3, a critical protein in the pathogenesis of fatty liver disease and fibrosis. Galectin-3 plays a major role in diseases that involve scarring of organs including fibrotic disorders of the liver, lung, kidney, heart and vascular system. The drug binds to galectin-3 proteins and disrupts its function. Preclinical data in animals have shown that GR-MD-02 has robust treatment effects in reversing liver fibrosis and cirrhosis. GR-MD-02 also has robust efficacy in pre-clinical cancer models in combination with immunotherapy agents.
About Galectin Therapeutics
Galectin Therapeutics is dedicated to developing novel therapies to improve the lives of patients with chronic liver and cancer. Galectin's lead drug (GR-MD-02) is a carbohydrate-based drug that inhibits the galectin-3 protein that is directly involved in multiple inflammatory, fibrotic, and malignant diseases. The lead development program is in non-alcoholic steatohepatitis (NASH) with cirrhosis, the most advanced form of NASH related fibrosis. This is the most common liver disease and is believed to be one of the largest drug development opportunities available today. Additional exploratory development programs are in combination immunotherapy for advanced melanoma and other malignancies. Galectin seeks to leverage extensive scientific and development expertise as well as established relationships with external sources to achieve cost-effective and efficient development. Additional information is available at www.galectintherapeutics.com.
About
About RECIST Criteria
RECIST is a set of published rules that define when tumors in cancer patients improve ("respond"), stay the same ("stabilize"), or worsen ("progress") during treatment. The criteria were published in
Further information on RECIST criteria is available at: https://ctep.cancer.gov/protocolDevelopment/docs/recist_guideline.pdf
Forward Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements relate to future events or future financial performance, and use words such as “may,” “estimate,” “could,” “expect” and others. They are based on management’s current expectations and are subject to factors and uncertainties that could cause actual results to differ materially from those described in the statements. These statements include those regarding the hope that Galectin’s development program for GR-MD-02 will lead to the first therapy for the treatment of fatty liver disease with cirrhosis and those regarding the hope that our lead compounds will be successful in cancer immunotherapy. Factors that could cause actual performance to differ materially from those discussed in the forward-looking statements include, among others, that the current management leadership may not be as effective as the predecessor management team; for the clinical trials in cancer immunotherapy, Galectin has relied on the trials undertaken by Providence, which limits the number of patients included in the trials; Galectin may be unsuccessful in expanding the scope of the cancer immunotherapy trials, and the results of expanded trials may not be positive; Galectin may not be successful in developing effective treatments and/or obtaining the requisite approvals for the use of GR-MD-02; manufacturing of drug product now in scale-up may not be successful or meet regulatory expectations, the Company’s Phase 3 clinical trial for the treatment of fatty liver disease, now in the initial planning stages, and any future clinical studies, including those in connection with cancer immunotherapy may not proceed and may not produce positive results in a timely fashion, if at all, and could prove time consuming and costly; plans regarding development, approval and marketing of any of Galectin’s drugs are subject to change at any time based on the changing needs of the Company as determined by management and regulatory agencies; regardless of the results of any of its development programs, Galectin may be unsuccessful in developing partnerships with other companies or raising additional capital that would allow it to further develop and/or fund any studies or trials. Galectin has incurred operating losses since inception, and its ability to successfully develop and market drugs may be impacted by its ability to manage costs and finance continuing operations. For a discussion of additional factors impacting Galectin’s business, see the Company’s Annual Report on Form 10-K for the year ended
KEYTRUDA® is a registered trademark of
Investor Contact:
Jack Callicutt, Chief Financial Officer
Media Contact:
Gregory FCA
Leigh Minnier, Vice President
610-228-2108
leigh@gregoryfca.com
A photo accompanying this announcement is available at http://www.globenewswire.com/NewsRoom/AttachmentNg/6ec20cf5-0917-4492-afe8-a399f7836703
Source: Galectin Therapeutics Inc.